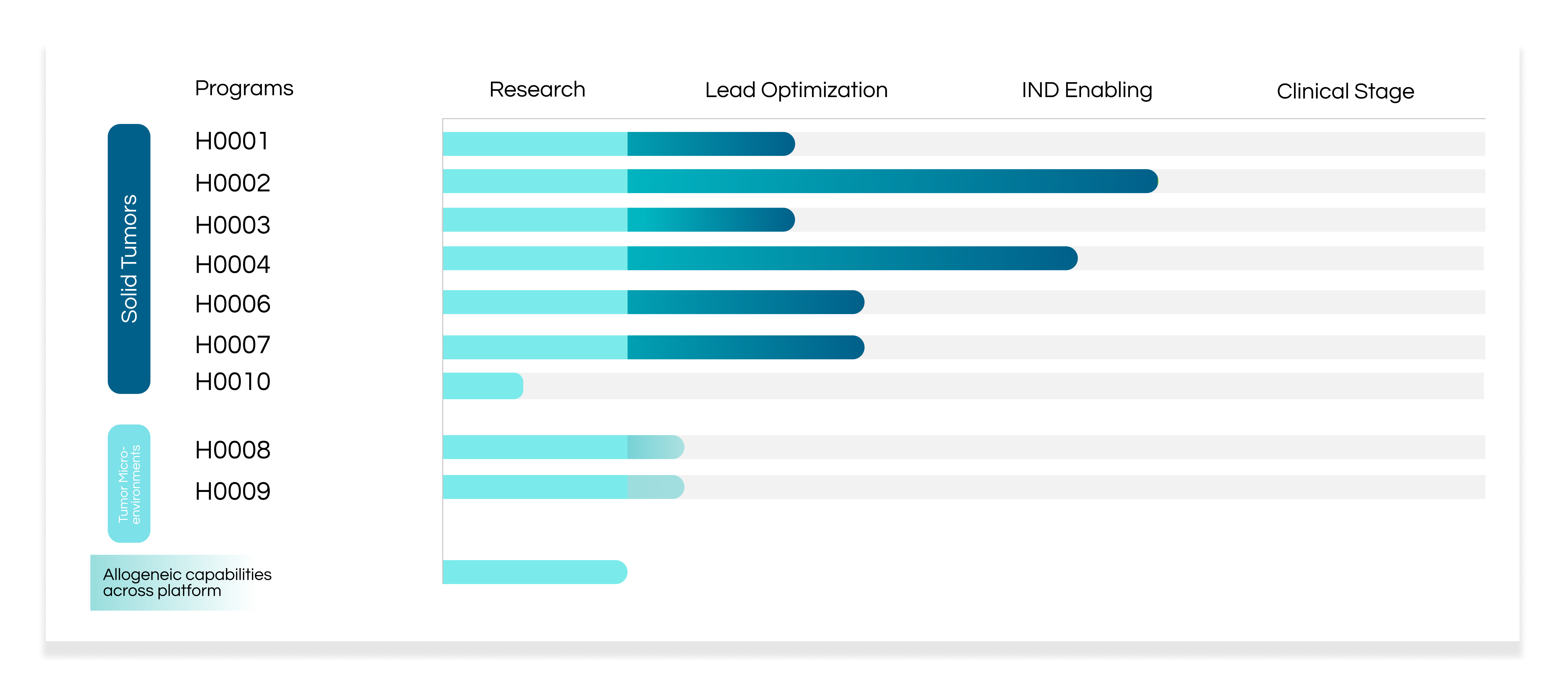
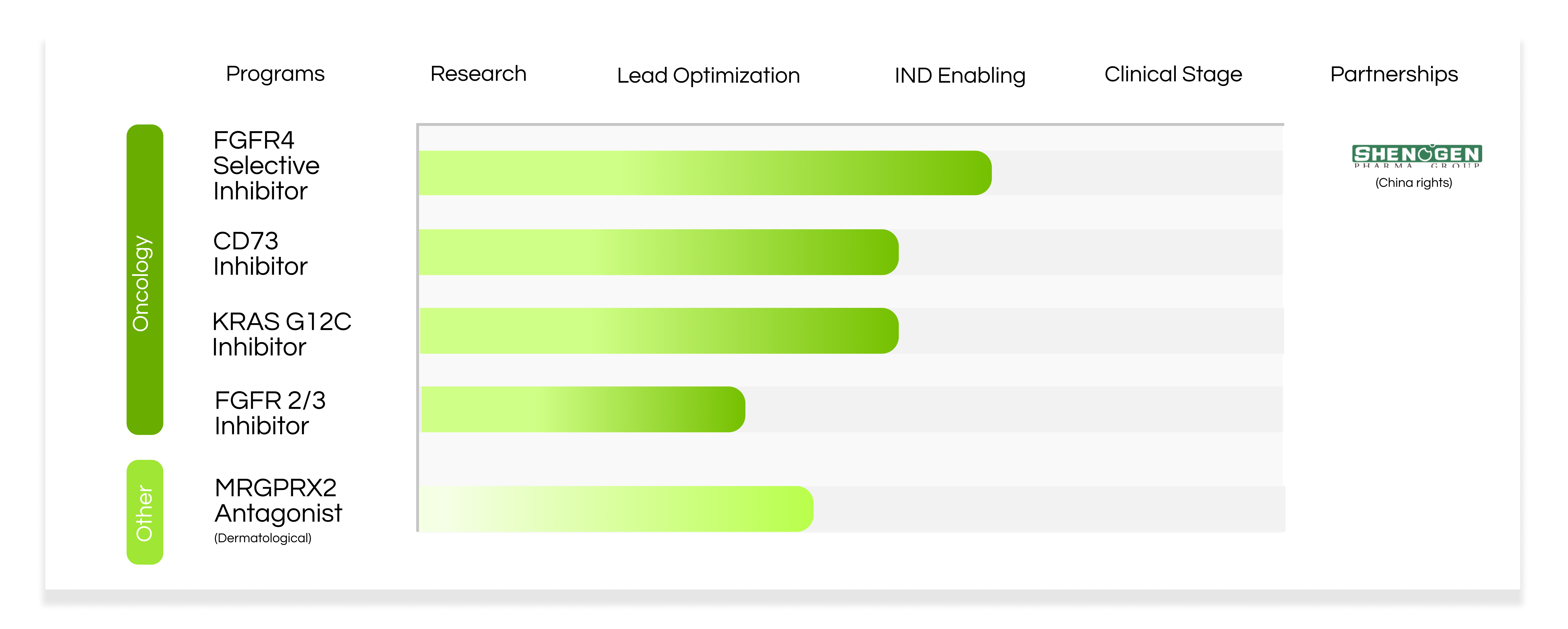
BioArdis’s portfolio of therapies includes a pipeline of small molecule therapies that are largely focused on oncology and immuno-oncology drug candidates, and a pipeline of CAR-T therapies targeting solid tumors.
Fibroblast growth factor receptor 4 (FGFR4) is a tyrosine kinase receptor that selectively binds fibroblast growth factor 19 (FGF19). Aberrant FGF19 signaling through FGFR4 has been implicated in the development and progression of a variety of cancers, in particular hepatocellular carcinoma (HCC) 1,2,3
BioArdis has developed a novel series of FGFR4 inhibitors whose potency and efficacy in liver cancer models has been demonstrated to be superior to other known clinical candidates.
CD73 is the rate-limiting enzyme in the ADO pathway and determines the extent of immune dysfunction in the tumor microenvironment (TME). CD73 expression is upregulated in the TME. CD73 has distinct roles in hematopoietic and non-hematopoietic cells to regulate anti-tumor T-cell response. Preclinical studies have demonstrated the therapeutic potential of targeting CD73 with monoclonal antibodies (mAbs) pharmacological inhibitors or siRNA. If intracellular CD73 activity is important for tumor growth, small molecules should be advantageous over mAbs and several companies have disclosed small-molecule inhibitor programs. 1,2,3,4
KRas is the single most frequently mutated oncogene and first gene to be causally implicated in human cancer . Mutations in KRas are prevalent amongst the top three most deadly cancer types in the United States: pancreatic (95%), colorectal (45%), and lung (35%).
Formerly considered an ‘undruggable’ target due to decades of failed approaches to directly targeting RAS and lack of efficacy with indirect (e.g. FTI) approaches. However, recent demonstration of covalent inhibition of a single driver mutation G12C (causal in 14% lung adenocarcinomas and 5% CRC adenocarcinomas) has led to renewed interest in this high-value target.
BioArdis has developed a novel series of KRas G12C inhibitors as a potential new treatment for patients with cancers bearing KRas G12C mutations.
Fibroblast growth factor receptors (FGFRs) are receptor tyrosine kinases with a role in several biological processes, such as the regulation of development and tissue repair. FGFR aberrations, particularly those affecting FGFR2 and 3, are common in a wide variety of cancers. Of 455,440 estimated patients who died of cancer in 2019, 17,019 (3.7%) were estimated to have FGFR2 or FGFR3 alterations. These data suggest that FGFR2/3 inhibition could be an important therapeutic option across multiple tumor types. 1,2,3
BioArdis is developing a novel series of FGFR2/3 inhibitors as potential new treatment for patients with cancers bearing FGFGR2 or FGFR3 alterations.
Itch, or pruritus, is the unpleasant sensation that leads to a desire to scratch. It is a common and distressing symptom in a variety of conditions and diseases. Currently the standard treatment for itch is the administration of antihistamines. However, not all forms of itch are responsive to antihistamines and there is no universally effective anti-pruritic drug. There is an urgent need for therapeutic agents for itch that do not target the histamine pathway.
The receptor MRGPRX2 is a member of a family of 5 human, 31 mouse, and 2 rat coding sequences that is related to the Mas1 oncogene. MRGPRX2 has been shown to have a limited expression profile, both peripheral and within the central nervous system, with highest levels in dorsal root ganglia and mast cells.
MRGPRX2 is activated by basic secretagogues – molecules known to activate mast cells through a non-IgE mechanism. Many drugs associated with systemic pseudo-allergies or anaphylactic reactions are MRGPRX2 agonists. Therefore, antagonists of human MRGPRX2 may be of benefit in the treatment of itch, inflammation, and drug reactions.
BioArdis has discovered small molecule MRGPRX2 antagonists that fill an unmet and immediate need for diseases caused by mast cell degranulation, such as anaphylaxis, pruritus, asthma and inflammatory pain. 1,2
BioArdis’s CAR-T platform is powered by a proprietary and highly differentiated large-scale screening engine that begins with target tumor antigen selection and optimizes key elements of a CAR to specifically target selected tumor antigen. Each of our candidates are autologous, single-target CAR-Ts, however we are actively developing allogeneic capabilities to ultimately treat patients with “off-the-shelf” therapies. Our discovery engine is also being utilized to develop next generation CAR-Ts that will overcome major obstacles against treating solid tumors, including antigen loss and inhibitory tumor microenvironments.